Instruments
Light Microscopy
Abberior MIRAVA POLYSCOPE

The Abberior MIRAVA POLYSCOPE is a 2D/3D STED with 2D MINFLUX. It is equipped with 6 excitation lines (405,440,485,561,640,685nm), two depletion lasers (589 ,775nm), RAYSHAPE (adaptive optics), MATRIX detector and FLIM capabilities.
Bruker/JPK NanoWizard Sense AFM & Nikon A1R Confocal

The Bruker NanoWizard Sense+ is an Atomic Force Microscope that probes surface topography and investigates mechanical properties of cells such as stiffness. AFM tips can be coated with various surface chemistries which can be used to study complex biological questions including receptor ligand interaction. The NanoWizard Sense+ is equipped with PeakForce Tapping which provided high-resolution imaging while simultaneously providing nanoscale property mapiing. This instrument also has DirectOverlay abilities allowing AFM imaging to be used in conjunction with a Nikon A1R confocal to image biological specimens. Additionally AFM can be perfomred in conjunction with widefield, TIRF and STORM microscopy.
This instrument was purchased with the generous support of the Robert H. Lurie Comprehensive Cancer Center via the Lea Charitable Trust, the Department of Cell and Developmental Biology, the Feinberg Dean's Office, and the Office for Research.
FlexCell Fx-6000T Tension System

The FlexCell Tension System uses vacuum and positive air pressure to apply cyclic or static strain to cultured cells. This can mimic physiological conditions found in blood vessels, muscles, lung, etc. Preset waveforms include: static, sinusoidal, heart stimulation, triangular and square. Custom waveforms can be programmed.
ImageXpress Confocal HT.ai Molecular Devices

This equipment enables high-throughput imaging including multiple modalities: phase contrast, brightfield imaging, widefield and confocal imaging with water immersion optics. It is equipped with an AgileOptix spinning disk technology. It allows multiplexing with 7 lasers and 8 imaging channels (DAPI, CFP, FITC, YFP, TRITC, Cy5, Cy7, TexasRed), and is equipped with a liquid dispenser, optimal for long-term imaging and high-throughput compound screening.
LaVision UltraMicroscope II Light Sheet

The LaVision light sheet UltraMicroscopes allow users to image cleared tissue in either aqueous buffers or organic solvents.There are six light sheets for homogenous illumination and the sheet width is 4-24um. It has four available laser lines: 488, 561, 637, 785. CAM has two body options available. The first is the zoom body which features an optical zoom from 1.26x to 12.6x and a working distance of up to 6mm. The second is the Super Plan option with infinity corrected optics. There are 3 different objectives (1.1x, 4x, 12x) which allows for 0.66x to 30x imaging. The working distance ranges from 10.9mm-17mm.
Leica DiveB SP8 Multiphoton
The Leica DiveB Sp8 Multiphoton is an upright multiphoton tailored for deep-tissue imaging. There are two tunable lasers: a Spectra Physics Mai Tai (690-1040)nm and a Spectra Physics InSight X3 (680-1300nm). It is capable to detecting fluorophores emitting from 380-780nm. There is also FLIM (fluorescent lifetime imaging) detection.
Nikon AZ100 Multi-Purpose Zoom Microscope

The AZ-100 covers a wide range of magnifications from 5X to 400X. It combines the advantages of stereo zoom microscopes and compound microscopes. The system can image blue, green and red-emitting fluorophores in thick tissues (e.g. Zebrafish, chicken eggs). The facility has obtained funding which will allow us to convert the system into a scanning confocal macroscope upon request. Please contact Dina for further details.
The AZ-100 was acquired through the support of Northwestern University Office for Research.
Nikon A1 Confocal Laser Microscope System

The A1 is a laser scanning confocal with four laser lines (405, 488, 561, 647). Available emission filters are 450/50, 525/50, 600/50 and 685/70. Four-color detection is possible with two standard PMTs (blue and far red) and two high-sensitivity GaAsP detectors (green and red). The system is equipped with the PerfectFocus focal drift compensation mechanism and automated XY stage.
The system was acquired through the generous support of Northwestern University Office for Research and Feinberg School of Medicine Dean’s Office. Additionally, several components of the system are on consignment from Nikon Instruments.
Nikon A1R+ Confocal Laser Microscope Systems (2 Systems)

The A1R is a laser scanning confocal with resonant scanning technology can reach 400 fps raster scan speed. We currently have two instruments available. The first system has six laser lines (408,458,488,515,561,638). Available emission filters are 450/50, 485/40, 525/50, 545/40, 600/50 and 650LP. Four-color detection is possible with two standard PMTs (blue and far red) and two high-sensitivity GaAsP detectors (green and red) as well as multispectral detectors for accurate emission separation.
The second system has four laser lines (408, 488, 561 and 638).Emission filters available are 450/50, 525/50, 600/50 and 685/70. Four-color detection is possible with two standard PMTs (blue and far red) and two high-sensitivity GaAsP detectors (green and red) for live cell or high-sensitivity applications. Both systems are equipped with the PerfectFocus focal drift compensation mechanism, automated XY stage and an incubated Tokai Hit chamber the A1R also serves as a powerful platform for multipoint live cell imaging.
The first system was acquired through the generous support of Northwestern University Office for Research and Feinberg Dean’s Office. The second system was purchased through the support of the Lurie Cancer Center and the Feinberg Dean’s Office.
Nikon A1RMP+ Multiphoton

The Nikon A1R-MP is an upright mulitphoton microscope tailored for deep-tissue imaging with penetration depth of up to 1-1.5mm, depending on the sample type. The tunable Chameleon Vision titanium sapphire laser can tuned within the range of 700-1000nm for excitation of most commonly used dyes within the blue and red emission range. It has GaAsP detectors for increased sensitivity. In addition, the system is equipped with resonant scanner, capable of scanning up to ~400 frames per second, thus making this an ideal system to perform intravital imaging, even on rapid biological processes.
The A1R-MP+ multiphoton microscope was acquired through an S10 shared instrumentation grant awarded to Teng-Leong Chew (1 S10 OD010398-01).
Nikon AXR Confocal (3 systems one with NSPARC)

The AXR is a laser scanning confocal with a 25mm field of view, galvano scanning up to 8192x8192 pixels, resonant scanning at 2048x2048 pixels that can scan as fast as 800 fps (small field of view). Two systems have four laser lines (408,488,561,640); the other has an addtional four lines (445,515, 594 and 750). Available emission filters are 450/50, two tunable emission band pass filters and 650LP. Four-color detection is possible with high-sensitivity GaAsP detectors. Multispectral detectors can also be performed for accurate emission separation. The system is also equipped with the PerfectFocus focal drift compensation mechanism, automated XY stage and an incubated Tokai Hit chamber. The AXR also serves as a powerful platform for multipoint live cell imaging. Additionally, the system software has AI image acquisition assistance to help users optimize capture settings and AI image denoise for higher quality images.
Lastly one of the two systems has the NSPARC detector. This arrayed detector has higher sensitivity, increased signal to noise and can almost double the resolution of the confocal.
The system was acquired through the generous support of Northwestern University Office for Research, Feinberg Dean’s Office and the Robert H. Lurie Cancer Center.
Nikon Biostation IMQ (2 Systems)

The Nikon Biostation IMQ is an integrated cell incubator and microscopy system that allows users to conduct long-term, multi-point live cell imaging. The Biostation facilitates a broad array of time-lapse experiments by providing environmental control of temperature, humidity and gas concentration with phase and fluorescence imaging (red and green). The first system holds 35 mm glass bottom dishes, and the second system is configured with a holder for chambered coverglass.
The first system was acquired through the generous support from Northwestern University Office for Research and Skin Biology & Diseases Resources-Based Center. The second system was purchased through the support of the Northwestern University Office for Research.
Nikon Biostation CT
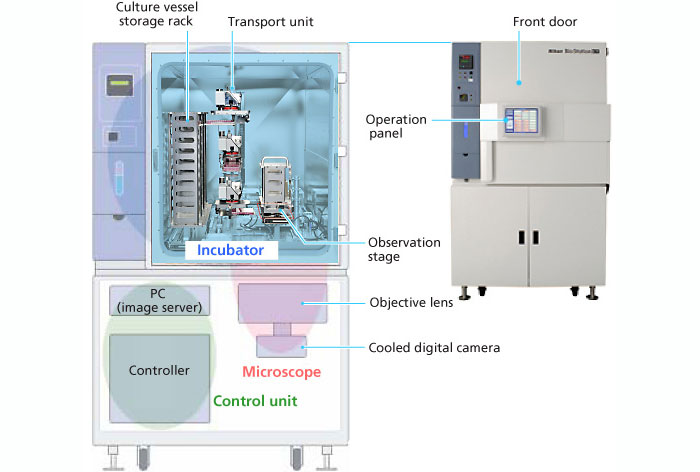
The Nikon Biostation CT is a high-content live-cell imaging system featuring a 30-pate stacker, robotic arm and integrated microscopy system within a full-size cell incubator. This allows users to conduct long-term, multi-point live cell imaging in a variety of formats, up to 96-well plates. The Biostation CT facilitates a broad array of time-lapse experiments by providing environmental control of temperature, humidity and gas concentration with phase and fluorescence imaging (blue, green and red).
The Biostation CT was acquired through an S10 shared instrumentation grant awarded to Joshua Rappoport (1 S10 OD021704 01A1).
Nikon W1 Dual Cam Spinning Disk Confocal with TIRF

This inverted microscope supports widefield, TIRF with a 95B prime Photometrics camera. There is also Yokogawa CSU-W1 spinning disk confocal imaging with two Hamamatsu Flash 4 cameras. Those imaging modalities can be combined with a Bruker Miniscanner capable of user-defined photobleaching and photoconversion experiments. The instrument is also equipped with a Nikon H-TIRF attachment to allow for total internal reflection microscopy. Long-term live cell imaging experiment is supported by an Tokai stage CO2 incubator and Nikon Perfect Focus Ti2 microscope. Available laser lines include 405, 445, 488, 514, 561 and 647.
Nikon CSU-W1 SoRa

This inverted microscope supports widefield, spinning disk confocal imaging and SoRa modes. It uses magnification and microlensing, together with deconvolution to achieve a lateral resolution of 120 nm. The instrument is equipped with 60x and 100x objective lenses for optimal resolution improvements. It employs an intermediate magnification of 2.8x for 100x and 4x for 60x to provide optimal effective pixel sizes for super-resolution and deconvolution. Long-term live cell imaging experiments are supported by a Tokai stage CO2 incubator and Nikon Perfect Focus Ti2 microscope stand. Available laser lines include 405, 488, 561 and 640.The Nikon SoRa was acquired through an S10 shared instrumentation grant awarded to Dina Arvanitis (1 S10 OD032270 01).
Nikon Ti2 Widefield (2 Systems)

Both Ti2 systems are equipped with LED light sources and filters for imaging blue (405), green (488), red (560) and far red (640) fluorescence. The Ti2 (A) is equipped with an LED-based Fura-2 illuminator. The Ti2 (B) has an additional 750 excitation line for imaging Cy7 based fluorophores. Both systems are capable of RGB color capture. Both have high-sensitivity, large-format cameras that results in high-resolution images that take advantage of the larger field of view. They have a variety of objectives allowing it to capture images from cells grown on plastic tissue culture plates as well as high-resolution images from samples imaged through coverglass. The microscope also has an automated stage for precise stitching and time-lapse imaging. Our facility has a heated, CO2-regulated incubation chamber that can be added to the Ti2 microscope to image live cell experiments.
The system was acquired through the generous support of Northwestern University Office for Research, Feinberg Dean’s Office and the Lurie Cancer Center.
Olympus DSU & Eppendorf Femtojet Microinjector

The Olympus DSU is a unique spinning disk microscope that comes equipped with three disks of varying slit width that giving users the capability to swap disks so that they are matched to magnification. The DSU uses arc lamp illumination for excitation wavelengths in the range of 350-700nm, allowing for imaging of reporters such as Fura dyes. The DSU also has a Tokai Hit chamber for live cell imaging as well as a Femtojet Microinjector that injects small to intermediate volumes (up to 100pl) of aqueous solutions into cells.
The Femtojet was generously donated by Sui Huang after being purchased by funding obtained by the Whitaker Fund. The Olympus DSU was acquired through the support of Northwestern University Office for Research.
Spectral Imaging Instruments Lago Bioluminescence/Fluorescence Imaging System

The LAGO from Spectral Imaging Instruments is capable of both in vivo bioluminescent as well as fluorescent imaging of small animals. Bioluminescent in vivo imaging is an exceptionally sensitive method of tracking cell migration and growth in vivo. Fluorescence animal imaging with 14 LED wavelengths from 360-805nm and 20 emission filters from 490-870nm and allows multiple channel acquisition. The LAGO allows imaging of up to 10 mice at time. It is housed in the pathogen free barrier in the Lurie Vivarium and accessible only by users with approved IACUC animal protocols. Users are encouraged to consult with CAM staff when designing their experiments.
The system was acquired through the generous support of Northwestern University Office for Research, Feinberg Dean’s Office and the Lurie Cancer Center.
TissueGnostics

TissueGnostics is a high-throughput, automated slide imaging system. It is capable of capturing large-format, high-resolution, stitched widefield images in either brightfield or four-color fluorescence from up to eight slides at a time. In combination with the user friendly HistoFaxs software package, scientists are able to quantify subpopulations of cells (i.e., double positive vs. total cells) and have the areas where these cells are present be highlighted in the images.
The TissueGnostics system was acquired through the support of Northwestern University Office for Research.
Zeiss PALM Laser Capture Microdissection System

The PALM system uses a unique combination of a high-precision microdissection laser and a catapulting pulse of defocused laser to effectively transport the specimen of the obejct plane into a collection device. The pressure catapulting mechanism eliminates heating of thermoplastic film. Specimen are usually mounted on a poly ethylene naphthalate (PEN) or polyethylene tetraphthalate (PET) membrane. This system is also able to dissect tissue mounted glass slides coated with a PEN membrane. It is capable of cutting sections up to 200um thick with 2-3um precision. This allows for live cell imaging and microdissection of subcellular structures.
The Zeiss PALM microdissection system is purchased through a National Center for Research Resources S10 Shared Instrumentation
Electron Microscopy
FEI Tecnai Spirit G2

The FEI Tecnai Spirit G2 is a 120kV transmission electron microscope (TEM). In addition to its greatly improved capability to generate high contrast and to image much thicker samples, the system is capable of performing 3D TEM tomography. This is a tomography technique for obtaining detailed 3D structures of subcellular macromolecular object. Using a tiltable sample holder, the sample is rotated, usually at 1 degree increment during the 3D image acquisition process, as an electron beam is therefore passed through the sample. The information is then collected and used to reconstruct a three representation image of the target. Current resolutions of 3D tomography are in the 5-20 nm range, suitable for examining supra-molecular multi-protein structures, although not the secondary and tertiary structure of an individual protein or polypeptide.
It was purchased with the support of Northwestern University Office for Research. Please contact Lennell for more details or training.
JEOL NeoScope SEM

The NeoScope benchtop SEM complements both optical microscopes and traditional SEMs in the lab and can be configured for advanced analytical applications. This compact electron microscope has the powerful electron optics of an SEM, with up to 60,000X magnification and offers both an Everhart Thornley type SE detector as well as high-sensitivity solid state BSE detector. The microscope has a sleek new design with up-to-date features. Operation is via a touch screen and is simplified with auto focus, auto alignment, auto contrast and auto brightness controls. The NeoScope operates in both low and high vacuum modes with three settings for accelerating voltage. These parameters are suitable for a variety of applications, all of which can be programmed in special pre-stored recipe files.
The system was acquired through the support of the Office for Research with cost share from the Feinberg Dean’s Office.
Leica EM ACE600 Sputter Coater

The Leica sputter coater EM ACE600 is equipped with a configurable metal process chamber. It is flexible and can be adapted to a wide range of applications. It uses a base vacuum of <2x10-6 mbar and fine balanced process parameters for a range of sputter targets. The sample distance and coating angle can be adjusted to the needs of the morphology of the sample. It ensures high transparency to electrons, adequate strength to withstand electron bombardment, and uniform thickness, key for quantitative imaging.
Leica EM TP Tissue Processor
The Leica EM TP tissue processor is used to prepare TEM samples. All sames in the same batch are identically prepared and the preprogrammed protocols allow for high reproducibility across batches.
Leica Freeze Substitution System

This unit can be programmed to perform stepwise, microprocessor-controlled changes in fixation conditions for user samples. It was purchased through the support of the Office for Research with cost share from the Lurie Cancer Center.
Leica EM UC6 and UC7 Ultramicrotomes

The Leica UC6 and UC7 ultramicrotomes have optimized stereo microscope positioning for specimen approach either with a diamond or glass knife to cut ultra-thin sections of specimens. They have motorized North-South movement of the knife stage, a motorized knife stage necessary for the E-W Measuring System and Autotrim Function automatic feed. They are ideal for biological application and soft materials preparation. They use Gravity Pull, an Integrated Antivibration System which prevents influence from external vibrations. Please contact Lennell for more details.
LKB Knifemaker

This knife maker allows users to create glass knives for electron and light microscopy sectioning. The thickness of glass used is 6-7mm. Please contact Lennell for additional information.