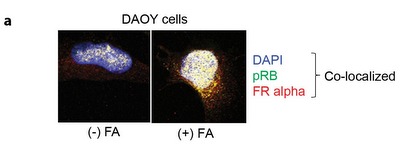
Cells were treated or not treated with folic acid and immunostained. Yellow signals indicate co-localization of folate receptor alpha (red) and pRB (green) in the nucleus.
Folic acid (FA) has traditionally been associated with prevention of neural tube defects; adequate amounts of FA in women before and during pregnancy are essential for the prevention of major birth defects. More recent work suggests that FA may also be involved in the prevention of adult onset diseases such as Alzheimer’s disease, dementia, neuropsychiatric disorders, cardiovascular diseases and cerebral ischemia.
Research spearheaded by Kyu-Won Shim and initiated by Vanda Boshnjaku from the Pediatric Neurosurgery Laboratory at Ann & Robert H. Lurie Children’s Hospital of Chicago Research Center highlights a novel role of folate receptor alpha (FRα) as a transcription factor (a protein that functions as a molecular switch, turning genes on or off). As the role of FA in human health and disease expands, it becomes increasingly critical to understand the mechanisms behind FA action.
This work examined the hypothesis that FRα acts as a transcription factor by binding to specific regions of DNA to control the activity of certain genes. FRα is a GPI-anchored protein that is attached to the cell surface and a component of the caveolae fraction. Disruption of FRα in mice may result in pups with a range of malformations and is lethal to the embryos at the time of neural tube closure. The studies show that FRα moves to the nucleus, where it binds to regulatory elements at promoter regions of two genes, fibroblast growth factor receptor 4 (Fgfr4) and Hes1, and regulates their expression. The research indicates that the FRα recognition domain maps to AT rich regions on the promoters.
These studies describe a novel role for FRα as a transcription factor, which has thus far been considered only as a folate transporter. The observation that FRα acts like a transcription factor is relevant to our understanding of the mechanisms of FA action during development and has significant implications for disorders associated with FA deficiency and FRα misregulation, and for management of human cancers that express FRα as a tumor antigen.
C. Shekhar Mayanil, PhD, senior author of the publication and a research associate professor in neurological surgery, is encouraged by the findings but acknowledges that more research is needed.
“This novel role of FRα as a transcription factor gives unique insight into developmental mechanisms associated with FA responsiveness. It also provides us with an exciting new avenue to explore for treatment of diseases associated with FA deficiency, FRα misregulation, and cancers that express FRα as a biomarker,” he says.
The paper, entitled “Nuclear localization of folate receptor alpha: a new role as a transcription factor” is available online in Scientific Reports, a Nature Publishing Group journal.
This work was supported by the State of Illinois Excellence in Academic Medicine award, a grant from the Spastic Paralysis Research Foundation of Illinois-Eastern Iowa District of Kiwanis, the Spina Bifida Association and Ann and Robert Lurie Children’s Hospital of Chicago Research Center pilot grant award.